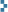Note: Click on any image for a larger version and a caption.
William J. A. Bailey (1884-1949) was a quack-cure huckster. After dropping out of Harvard without a degree, he briefly engaged in mail fraud, served a prison term, and then entered the lucrative and minimally-regulated patent medicine trade with a fraudulent European doctorate. His chosen specialty was “male enhancement.” (As anyone with an email account will attest, this dubious market has survived the intervening century and all attempts at regulation.) Bailey’s first boner pills contained strychnine. He entered business at a time when popular enthusiasm for radioactivity was ascendant, and he is mostly remembered today for his lethal radioactive quack cures, including Radithor and the Radiendocrinator (above). Most hucksters did not actually include radioactive ingredients in their products; they lied. On this matter, though, Bailey was deadly honest. Evidence suggests he used his own products, believed in them, and in all possibility, died from them (bladder cancer).
The Radiendocrinator is a credit-card-sized radium source of spectacular activity (originally 100 microcuries of Ra-226 and 150 microcuries of Ra-228) intended to be stuffed into a man’s jockstrap and worn “under the scrotum” for extended duration. Production spanned 1922-1929, and with prices set in the thousands of dollars (1929 basis), only the Jay Gatsby set could afford these gilded nut-roasters. Users were instructed to orient the wire-mesh window towards the skin to ensure maximum beta dose to shallow tissues. In measurements on my Radiendocrinator (and it must be noted that the Ra-228 is long gone now and only Ra-226 remains), the beta-gamma reading on a Fluke 451B ion chamber was 390 mR/h at 1/8 inch, and the gamma-only reading was 52 mR/h. It is not straightforward to extract a beta dose rate from such measurements, but assuming a correction factor of ~0.1 Gy/R (dependent on beta energy, source geometry, and ion chamber geometry), a total scrotal skin and gonadal dose rate of 30-40 mGy/h is probably not unreasonable. Far from causing a boost to male potency, wearing a Radiendocrinator according to the manufacturer’s instructions would have likely led to temporary sterility and, of course, elevated risk of cancer. In other words, it was a male contraceptive of sorts. As an unsealed radium source, the wearer’s clothing, nutsack, schlong, bedsheets, sexual partners, and probably anything in the vicinity would have been rendered contaminated by Pb-210, Po-210 and other radon daughters. Lord, what a mess.
Modern owners of these radioactive collectibles should be cautious about proper storage, as they are among the hotter of the classic quack radium cures. Most important is a hermetic container (e.g., a small dive box) to control radon daughters emitted from the source itself. The blue velvet-lined Radiendocrinator case is likely to be roaring with radon daughter activity as well, and should be kept separately in a bag or other sealed container. Shielding from the penetrating gamma radiation is strongly advised. 2-4 cm of lead is reasonably effective. The source and its case should only be handled with gloves and the source itself should NEVER be opened except in a radiochemical glovebox facility, as there is a grave risk of airborne radium alpha activity being liberated.
UPDATE: VARSKIN 4 model
The question of dosimetry from a Radiendocrinator continues to interest me because of how high the doses could potentially be from this particular device in its suggested mode of long-term use pressed against the skin. To provide more insight into the doses, I downloaded VARSKIN 4, a deterministic radiation transport tool developed for the US NRC often used to model beta doses to skin, and I modeled the geometry and source activity of a Radiendocrinator at the peak of its beta-emitting powers (which occurs when it is 3.5 years old). The model makes numerous assumptions, and some may not be very good:
- Source area is the Radiendocrinator’s front “window,” 6.23 cm long and 3.63 cm wide (measured).
- The source itself is 7 sheets of absorbent paper uniformly loaded with radium sulfate, 0.33 mm thick each, with a density of 0.55 g/cc. The paper’s density and thickness are a total guess. The number of source sheets is borrowed from Paul Frame’s online description of the innards of his device. Note: NEVER TAKE ONE OF THESE APART (unless, like Paul Frame, you have the facilities to handle a loose alpha source of this intensity). Initial activity of 100 μCi Ra-226 and 150 μCi Ra-228 were inferred from Kolb’s and Frame’s description in Living with Radiation: The First Hundred Years.
- At the time of peak beta intensity–when the source is 3.5 years old–it will contain the following important beta-gamma activities:
- Pb-214, 100 μCi
- Bi-214, 100 μCi
- Ac-228, 98.4 μCi
- Pb-212, 84.2 μCi
- Bi-212, 84.2 μCi
- Tl-208, 30.3 μCi
- Pb-210 and Bi-210 are omitted as they will not have had much opportunity to grow in at 3.5 years. Alpha emitters are omitted.
- There are two overlain sheets of 16-mesh woven metal screen composed of 0.009-inch wire that are interposed between the source material and the human target. VARSKIN does not model such geometries. I calculate a transparency of 53%, and assume the metal blocks 100% of intercepted beta particles and 0% of intercepted photons.
- There is a plastic sheet, probably nitrocellulose, over the front of the device that I model in VARSKIN as 0.5 mm thick with a density of 1.3 g/cc. This is a total guess.
- I assume a 1-mm gap between the source and skin.
- VARSKIN’s default skin dose averaging area is 10 sq. cm., in recognition of the US NRC’s current rule for computing shallow dose equivalent in 10 CFR 20.1201(c). I did not alter this in the calculation.
Results: In vintage condition (3.5 years old), the Radiendocrinator’s predicted shallow dose rate due to beta particles is 88 mGy/h, and with the gamma contribution added in is up to 91 mGy/h. Deep dose rate (from gamma contributions only) is 2.0 mGy/h. In the Radiendocrinator’s present condition, assuming the contributions of ingrown Bi-210 and the total decay of the Ra-228 chain, the beta-gamma shallow dose rate is 57 mGy/h, and the deep dose rate is 0.9 mGy/h. So…what does this mean, practically, for the wearer?
- 2 Gy is the threshold for skin erythema: waves of redness and itching sensation over several months, culminating in skin death and replacement as in a sunburn. The Radiendocrinator wearer potentially earns an itchy, inflamed scrotum with a few nights of wearing the device.
- 15 Gy marks the onset of painful burning with moist desquamation following browning of the skin, i.e. a “nuclear tan”, with the possibility of long-lasting ulceration. This is a hardcore radiation burn. If you wore the Radiendocrinator all the time, every day, for a week, this might be your reward. As there are no records of gruesome and agonizing injuries associated with the device, I assume there were no users hardcore enough to “ride the radium” full-time.
- Temporary sterility can happen with doses of 150 mGy or greater to the testes. With a deep dose rate of 2 mGy/h, it would take a guy three whole days on the nuclear pad to achieve temporary sterility. Libido would not be impacted.
- Stochastic effects: using ICRP weighting factors, I calculate an effective dose rate of about 1.2 mSv/h from the skin (shallow) and deep (general tissue) dose rates given above. The excess risk of fatal cancer is on the order of 5%/Sv. Though the dose rate is on the higher side, your real problem with this source is the skin damage you would endure.